Percutaneous Vertebral Augmentation for Osteoporotic Vertebral Fractures: Overview of Early Influential Studies, Review of the Randomized Placebo-Controlled Trials and Commentary on Its Utilization in Clinical Practice
Jose J. Santiago, MD and Sahil V. Mehta, MD
The authors report no conflict of interest related to this work.
©2019 by The Orthopaedic Journal at Harvard Medical School
BACKGROUND ercutaneous vertebroplasty is a minimally invasive, image-guided procedure used as a treatment option for painful vertebral compression fractures in patients who fail conservative medical therapy. Several observational studies and open-label randomized control trials have provided data suggesting that vertebroplasty is beneficial in improving pain and quality of life, but its efficacy remains a point of contention. Studies published over the last decade have reported heterogeneous results that make it difficult to ascertain whether vertebroplasty truly provides benefit to patients. This narrative review offers a chronologic overview of the published research relevant to the treatment of osteoporotic vertebral compression fractures with vertebral augmentation.
METHODS A search for relevant articles published on PubMed and Cochrane electronic databases prior to December 2018 was performed using the following keywords (combined with appropriate Boolean connectors): “vertebroplasty”, “kyphoplasty”, “vertebral augmentation”. Randomized controlled trials, systematic reviews, meta-analyses, and cohort studies were screened for inclusion.
RESULTS PTwenty-nine publications were included in this narrative review: fifteen randomized controlled trials, eight systematic reviews and meta-analyses, and six prospective or retrospective cohort studies.
CONCLUSION The efficacy of vertebral augmentation in the treatment of osteoporotic vertebral fractures continues to be in question. Many of the most cited randomized placebo-controlled trials showed no clinically significant benefits of treatment with respect to pain, disability, or overall quality of life. When rigorous selection criteria are applied, vertebral augmentation remains a treatment option that may improve pain, result in earlier hospital discharge, and reduce morbidity in selected patients.
ABBREVIATIONS VCMT= Conservative Medical Therapy; EQ-5D=EuroQol five dimensions questionnaire; NRS=Numeric Rating Scale pain; QUALEFFO=Quality of Life questionnaire of the European Foundation for Osteoporosis; RDQ=Roland-Morris Disability Questionnaire; RMDQ= Roland-Morris Disability Questionnaire; VA= Vertebral Augmentation; VAS=Visual Analog Scale pain; VCF = Vertebral Compression Fracture
LEVEL OF EVIDENCE Level V Narrative Review
KEYWORDSVertebroplasty, kyphoplasty, vertebral augmentation, vertebral compression fracture
Approximately 10 million people in the United States have osteoporosis,1,2 and this large and growing demographic is at high risk for fragility fractures, particularly vertebral compression fractures (VCF). In the absence of severe deformity or spinal instability compromising the neurologic elements, vertebral fractures are typically treated with conservative medical therapy (CMT) with activity modification, analgesic medications, and potential use of a back brace.3 Patients are often in significant pain and require opioid analgesics and/or hospitalization.4 Decreased patient mobility, reduced lung aeration, and overall poor functional status place these patients at higher risk for pneumonia, venous thromboembolism, decreased independence and quality of life, and increased mortality.4
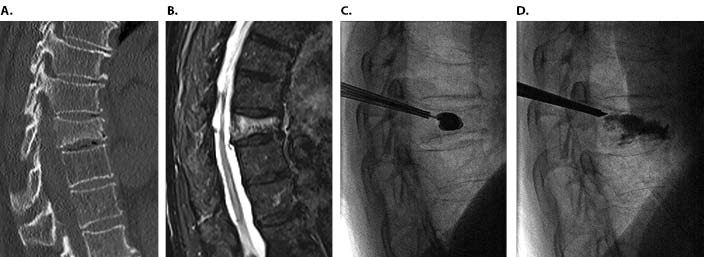
Since its introduction to the United States in the 1990s, percutaneous vertebral augmentation (first vertebroplasty and later balloon kyphoplasty) has been used as a treatment option for painful and debilitating vertebral compression fractures in patients who fail CMT.5-8 Percutaneous vertebroplasty involves image-guided injection of polymethylmethacrylate (PMMA) into the collapsed vertebral body from a posterior incision.6 Kyphoplasty involves the creation of a cavity within the collapsed vertebral body to restore vertebral height using a small balloon or curette prior to the injection of PMMA (Figure 1). Newer iterations of the procedure include implantable devices to restore height combined with cement augmentation.9 The goals of these procedures are to reduce pain, improve mobility, and improve quality of life.
Numerous early observational studies and several open-label randomized control trials (RCT) provided support for vertebral augmentation in improving pain and quality of life for patients with vertebral insufficiency fractures.11,12 Studies published over the last decade have reported mixed results, which makes it difficult to ascertain whether vertebral augmentation truly provides benefit to a specific group of patients.19,20,24,26,27 The efficacy of this treatment continues to be a point of contention.
The primary purpose of this article is to provide a chronologic overview of the published research relevant to the treatment of osteoporotic vertebral compression fractures with vertebral augmentation while highlighting the contentious findings.
A search for relevant articles published on PubMed and Cochrane electronic databases prior to December 2018 was performed using the following keywords (combined with appropriate Boolean connectors): “vertebroplasty”, “kyphoplasty”, “vertebral augmentation”. Inclusion criteria were randomized controlled trials, cohort studies, systematic reviews, and meta-analyses of vertebral augmentation procedures published in English. There was more emphasis on studies comparing the efficacy of vertebroplasty to conservative medical therapy for VCF, as no placebo-controlled RCTs for kyphoplasty have been published at the time of this review. Exclusion criteria include non-English publications, preliminary data from unpublished research, case reports, case series, letters, comments, studies of vertebral augmentation for traumatic VCF, studies comparing efficacy of kyphoplasty versus vertebroplasty, studies involving implants or other types of vertebral augmentation, and citations with substantial content redundancy. Given the narrative nature of this article, the included publications were not screened with the rigor of a systematic review, and no formal quality assessment was performed.
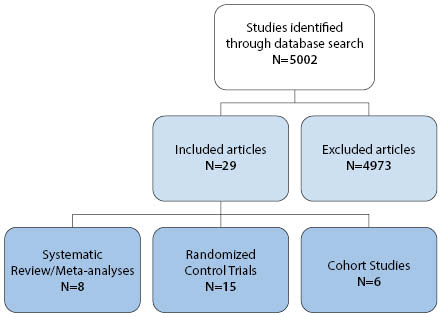
The search resulted in a total of 5,002 citations. After duplicates were removed, the articles were screened for inclusion according to the above criteria.
Twenty-nine publications were included in this review: fifteen randomized controlled trials (including four sham-controlled RCTs), eight systematic reviews and meta-analyses, and six retrospective and prospective cohort studies (Figure 2).
Observational studies
The first reported percutaneous vertebroplasty was performed in France in 1984 to fill a vertebral body void left by a hemangioma.10 In 1997, Jensen et al. published the first paper on percutaneous vertebroplasty in the United States after successful treatment of twenty-nine patients with forty-seven osteoporotic thoracolumbar compression fractures over a period of three years.5 Twenty-six of the twenty-nine patients (90%) reported pain relief and improved mobility within twenty-four hours of treatment with two patients reporting immediate pain relief. Other observational studies and case series have reported similar results.11 In 2006, Hochmuth et al. published a critical review of studies of percutaneous vertebroplasty for treatment of 2,086 patients with VCF from 1989 to 2004.11 Nineteen studies (out of thirty) reported pain measures using a visual analog scale (VAS) and showed significant pain relief for patients after vertebroplasty with a mean VAS before treatment of 8.1 (range 6.4-9.7), and after treatment of 2.6 (range 1.7-3.9), reflecting a 67.9% improvement (p<0.001).
A systematic review by Bouza et al. in 2006 included 1,710 patients from twenty-six early observational studies of percutaneous balloon kyphoplasty (twenty-one non-comparative and five comparative).12 Despite the heterogeneity between the study groups, patients who underwent kyphoplasty had more significant pain relief (VAS) compared to the medical treatment group, with a weighted mean difference of −5.11 (95% CI -5.72 to -4.49). Patients with osteoporotic vertebral compression fractures who underwent balloon kyphoplasty had an 8.6% to 40.6% increase in height of the compressed vertebral body with reduced local kyphosis.
In 2007, based on data from observational studies, several specialized medical societies published a consensus statement endorsing vertebral augmentation as an appropriate therapy for the treatment of painful vertebral compression fractures refractory to medical therapy when performed for the indications supported by the referenced literature.13Unblinded randomized controlled trials
The apparent benefits associated with percutaneous vertebroplasty seen in observational studies were reinforced with the publication of the VERTOS trial in 2007.14 VERTOS was the first randomized controlled trial comparing percutaneous vertebroplasty with optimal conservative treatment for VCFs. Thirty-four patients with subacute and chronic (six to twenty-four week old) VCFs, confirmed by radiography and MRI, were enrolled. Patients were at least fifty years of age with debilitating back pain and focal tenderness refractive to CMT. Of thirty-four patients, eighteen were randomized to vertebroplasty and sixteen to conservative medical treatment. Pain evaluation with a visual analog scale (VAS) obtained at baseline and one day after treatment showed statistically significant reduction in pain in the vertebroplasty treatment group compared to CMT. The mean VAS score decreased 2.4 points from baseline in the vertebroplasty group (from 7.1 to 4.7) and 0.5 points from baseline in the CMT group (from 7.6 to 7.1). After two weeks of treatment, 88% of patients in the medical treatment group crossed over to vertebral augmentation. Baseline and two week post-treatment Quality of Life Questionnaire of the European Foundation for Osteoporosis (QUALEFFO) and Roland-Morris Disability Questionnaire (RMDQ) were also collected. VERTOS demonstrated more significant pain relief and improvement of mobility, function, and stature in the vertebroplasty group compared to CMT in the as treated analysis.
Published in 2010, VERTOS II was a multicenter, open-label randomized trial comparing the efficacy of vertebroplasty to conservative medical therapy for VCF.15 Patients were at least fifty years of age with acute (less than six weeks old) compression fractures at T5 or lower level, as assessed by MRI. Of 202 enrolled patients, 101 received vertebroplasty and 101 underwent CMT. The study showed significant improvement in pain measured by VAS in the vertebroplasty group compared to CMT. The change in mean VAS score between baseline and 1 month after vertebroplasty was -5.2 (95% CI -5.88 to -4.72) and for CMT -2.7 (95% CI -3.22 to -1.98). One year after vertebroplasty, the change in mean VAS score was -5.7 (95% CI -6.22 to - 4.98) and for CMT -3.7 (95% CI -4.35 to -3.05). The difference between groups in reduction of mean VAS score from baseline was 2.6 (95% CI 1.74 to 3.37, p<0.0001) at 1 month and 2.0 (95% CI 1.13–2.80, p<0.0001) at 1 year. Quality of life was measured with the Quality of Life Questionnaire of the European Foundation for Osteoporosis (QUALEFFO), and physical function measured with the Roland Morris Disability (RMD) questionnaire. QUALEFFO and RMD scores at baseline were worse for the vertebroplasty group. However, improvement with time was significantly greater and quicker after vertebroplasty than with CMT, for both QUALEFFO (p<0.0001) and RMD (p<0.0001).
A 2011 randomized control trial by Farrokhi et al. comparing vertebroplasty with CMT enrolled eighty-two patients.16 Forty patients underwent vertebroplasty and forty-two received CMT.16 Significantly greater improvement in mean VAS score was reported in the vertebroplasty group compared to CMT at 1 week with a difference of -3.1 (95% CI -3.72 to -2.28; p<0.001). In addition, the study reported a mean gain in vertebral body height of 8 mm with associated mean reduction of kyphosis by 8° in the vertebroplasty group.16
In 2012, Blasco et al. published a randomized controlled trial comparing percutaneous vertebroplasty with medical therapy in patients with VCF.17 They enrolled 125 patients, with sixty-four undergoing vertebroplasty and sixty-one receiving CMT. Patients were clinically assessed with quality of life measurements (QUALEFFO) and VAS at baseline, two weeks, two months, six months, and twelve months. Both groups had improved VAS scores at all time points, but the vertebroplasty group had greater pain relief compared to CMT at two months (VAS difference compared to initial visit 3.07 ± 0.45 versus 1.59 ± 0.42, p = 0.017). Quality of life scores improved throughout the study in the vertebroplasty group, but no significant improvement was seen in the CMT group until the six month follow-up. The groups had similar pain relief and quality of life measures at twelve months.17
Balloon kyphoplasty also showed promising outcomes in early trials. The first randomized control trial comparing efficacy and safety of percutaneous balloon kyphoplasty to conservative medical therapy of osteoporotic vertebral compression fractures (FREE trial) was published by Wardlaw et al. in 2009.18 FREE was a multicenter study involving twenty-one sites in eight countries and enrolled 300 patients. Inclusion criteria were: at least twenty-one years of age, at least four out of ten pain on the numeric rating scale (NRS), between one and three acute or subacute fractures from T5-L5, focal tenderness, and magnetic resonance imaging (MRI) showing bone marrow edema. Balloon kyphoplasty was performed in 149 patients while 151 patients received CMT. Differences in pain associated with the vertebral fracture(s) were measured with the Short Form 36 (SF-36) physical component summary (PCS) score at baseline and one month after treatment. Safety and quality of life metrics were followed for up to one year. The study was completed by 138 of 149 patients (93%) in the kyphoplasty group and 128 of 151 patients (85%) in the conservative treatment group. There was more significant improvement in pain based on PCS score in the balloon kyphoplasty group (7.2 points from baseline; 95% CI 5.7–8.8) than in the CMT group (2.0 points from baseline; 95% CI 0.4–3.6). The balloon kyphoplasty group also showed a more significant improvement in quality of life based on EQ-5D questionnaire and Roland–Morris scores, which improved by 4.0 points (p < 0.0001) in the balloon kyphoplasty group and 2.6 points (p < 0.0001) in the medical therapy group. Clinically important differences as estimated for several quality of life measures were used when considering the clinical significance of outcomes, as they may differ from statistical significance. Clinically important differences range between 2 to 3 points for the Roland–Morris scale; 3.5 to 4.3 points for the SF-36 PCS; 1.0 to 2.5 points for back pain; 15 points for the SF-36 physical function score; and 0.08 for EQ-5D.18
Randomized placebo-controlled trials
Four randomized placebo-controlled trials using a sham procedure have been published (Table 1), and others are ongoing. In 2009, The New England Journal of Medicine (NEJM) published the first two randomized placebo-controlled trials comparing percutaneous vertebroplasty to a sham procedure.19,20 The INVEST study enrolled 131 patients who were at least fifty years of age with reported pain of at least three on the NRS, fracture age less than one year, and one to three osteoporotic vertebral compression fractures between T4-L5 confirmed by MRI or radiography. Sixty-eight patients were randomized to percutaneous vertebroplasty and sixty-three to sham treatment. The sham treatment consisted of an injection of 1% lidocaine in the tissues overlying the pedicles and a periosteal injection of the pedicles with 0.25% bupivacaine. Vertebroplasty was simulated by mixing the PMMA and by applying manual pressure over the spine. This study revealed no significant difference between the vertebroplasty and placebo group in the primary endpoint of back pain by NRS and RDQ scores one month after treatment: NRS difference, 0.7; 95% CI, −0.3 to 1.7; p=0.19; RDQ difference, 0.7; 95% CI −1.3 to 2.8; p=0.49.19
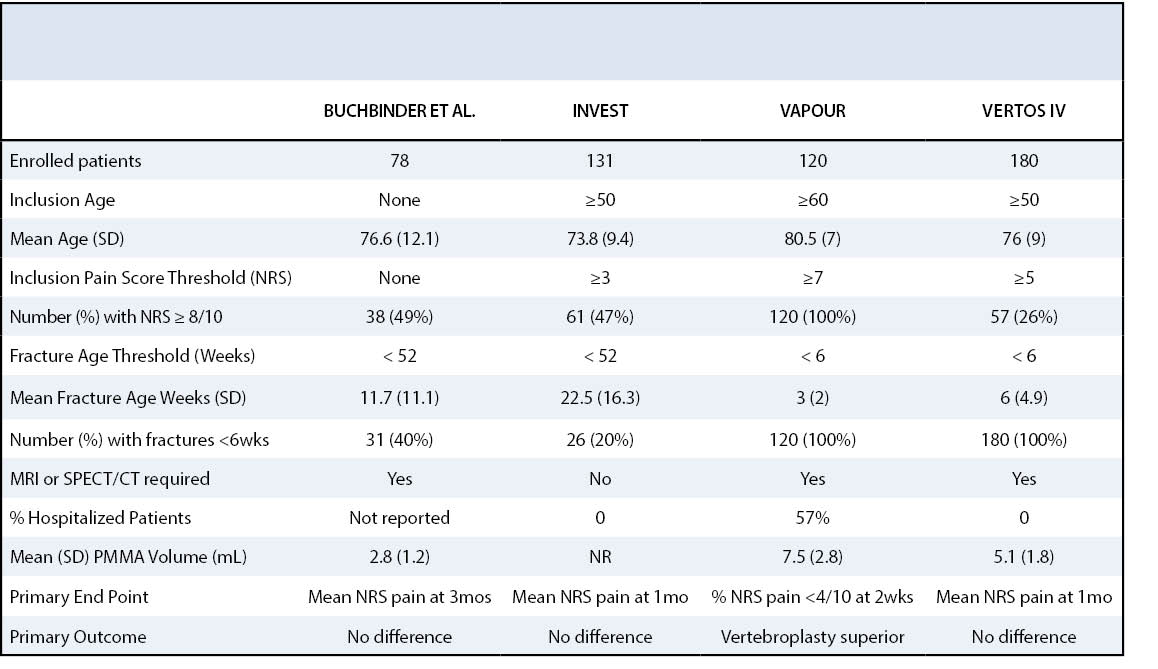
A second randomized placebo-controlled trial comparing percutaneous vertebroplasty to a sham treatment was published in the NEJM by Buchbinder et al. in the same year.20 The study enrolled seventy-eight patients with osteoporotic vertebral compression fractures confirmed by radiography and MRI with back pain of less than one year’s duration. No age criterion was specified. Thirty-eight patients were randomized to vertebroplasty and forty to sham procedure. To more accurately simulate vertebroplasty in the sham treatment, a needle was advanced to the periosteum then replaced by a blunt ended stylet, which was gently tapped with a hammer. Pain assessment was completed at baseline and at various intervals for two years after the intervention20 with the primary endpoint being pain at three months after intervention. Like INVEST, the trial by Buchbinder et al. showed no significant difference in pain, disability, or other quality of life measures between the vertebroplasty and sham treatment groups.
The results of these trials are in sharp contrast to the positive outcomes seen in the multitude of previous observational studies and unmasked randomized controlled trials.7,8,21 Critics of these studies, particularly of the trial by Buchbinder et al., point to the heterogeneity of the patient population with significant variability in the chronicity of the fractures and the lack of specified physical examination findings as inclusion criteria.8 Another issue relates to the potential confounding effect of the periosteal anesthetic injection used in the sham procedure.22,23
Two additional randomized placebo-controlled trials comparing vertebroplasty to a sham treatment have been published: VAPOUR in 2016 and VERTOS IV in 2018. VAPOUR was a multicenter, randomized, double-blinded, placebo-controlled trial on the safety and efficacy of vertebroplasty for acute painful osteoporotic fractures.24 In the study design, the authors aimed to address some of the limitations and points of contention of the aforementioned studies, including patient heterogeneity and confounding from therapeutic effect of periosteal anesthetic injection.22,23 The study focused on older patients with recent fractures and significant pain—a subgroup of patients that was anticipated to benefit from treatment.25The study included patients at least sixty years of age with reported pain of at least seven out of ten from a fracture less than six weeks prior that is confirmed by MRI or single photon emission computed tomography (SPECT). Over a study period of three years, 120 patients were enrolled with sixty-one randomized to vertebroplasty and fifty-nine to sham treatment. The primary endpoint was pain reported as less than four on the NRS at two weeks. The study showed a significant difference in pain relief between the groups, particularly when the intervention occurred within three weeks of fracture. Twenty-four (44%) of the patients in the vertebroplasty group and twelve (21%) in the control group had adequate pain relief (p=0·011). Quality of life measures (QUALEFO) were also better in the vertebroplasty group at 48 compared to the sham treatment group at 54 (p<0.03).24 The VAPOUR trial is the only sham-controlled trial of vertebroplasty to date that supports early intervention for a select group of patients with severe pain and acute fractures when the intervention occurred within three weeks of fracture.The most recent randomized placebo-controlled trial is VERTOS IV, published in 2018.26 This multi-center study enrolled 180 patients with acute VCFs at 4 centers. Ninety-one patients were randomized to percutaneous vertebroplasty, and eighty-nine were randomized to sham treatment. The primary endpoint of the study was mean reduction in VAS at one day, one week, one month, three months, six months, and twelve months. Clinically significant pain relief was defined as reduction in VAS score by at least 1.5 from baseline. The study showed a statistically significant reduction in pain in both groups, however, no significant difference in pain was demonstrated between groups at all timepoints: mean difference in VAS 0.20 (95% CI -0.53 to 0.94) at baseline, -0.43 (95% CI -1.17 to 0.31) at one day, -0.11 (95% CI -0.85 to 0.63) at one week, 0.41 (95% CI -0.33 to 1.15) at one month, 0.21 (95% CI -0.54 to 0.96) at three months, 0.39 (95% CI -0.37 to 1.15) at six months, and 0.45 (95% CI -0.37 to 1.24) at twelve months. Additionally, there was no significant difference between the groups in quality of life measures during twelve month follow-up.26 Three post hoc observations during follow-up found a statistically significant proportion of participants with VAS scores of five or higher at twelve months for the sham procedure compared with vertebroplasty (41% vs 20%). Follow up imaging showed a significantly higher incidence of vertebral body collapse after the sham procedure, which supports the assumption that vertebroplasty protects against progressive collapse of the vertebral body. VERTOS IV used periosteal local anesthetic infiltration; whereas VAPOUR only infiltrated the subcutaneous space. The therapeutic effect of periosteal infiltration of anesthetic is unclear and confounds the observed difference of the sham procedure group.
Hospital Admission and Mortality
An analysis of 13,624 hospitalized patients with VCFs from the French Hospital National Database demonstrated that a higher proportion of patients are discharged in seven days after vertebroplasty compared with medical management (68% versus 47%, P < .001).27 A large Taiwanese study using National Health Insurance data for 9,238 hospitalized patients with VCFs demonstrated discharge two days earlier and lower thirty day readmission rates after vertebroplasty.27
With the heterogeneity of results in numerous studies, it can be difficult to ascertain whether vertebroplasty provides benefit to patients. In clinical practice, many of those who have performed vertebroplasty will speak to its anecdotal benefits and pain relief. Reconciling these anecdotal results with clinical evidence is difficult. The NEJM studies from 2009 are often seen as a turning point in the life cycle of vertebral augmentation. Prior to these, augmentation had been utilized unchecked by some practitioners without high quality evidence. The external validity of these studies may be limited by the selection criteria including a heterogeneous group of patients comprising acute, subacute, and chronic fractures. Lack of MRI confirmation may have led to potentially missing adjacent radiographically occult non-VCF fractures in patients with significant pain and physical exam findings mimicking VCFs.
Vertebral augmentation may play a role in reducing pain, shortening hospital stays, and potentially decreasing mortality.28,30 It is telling that vertebral augmentation is supported by the Society of Interventional Radiology (SIR), American Association of Neurological Surgeons (AANS) and the Congress of Neurological Surgeons (CNS), American College of Radiology (ACR), American Society of Neuroradiology (ASNR), American Society of Spine Radiology (ASSR), Canadian Interventional Radiology Association (CIRA), and the Society of NeuroInterventional Surgery (SNIS), with the societies’ consensus statement that “the use of vertebroplasty or kyphoplasty is a safe, efficacious, and durable procedure in appropriate patients with symptomatic osteoporotic and neoplastic fractures, when performed in a manner in accordance with published standards.”31 However, counter to this statement, a report commissioned by the American Society for Bone and Mineral Research (ASBMR) concludes that "routine use of vertebral augmentation for pain relief after vertebral fracture is not supported by current data,” and that, if offered, “patients should be fully informed about the evidence”.32
The efficacy of vertebral augmentation in the treatment of osteoporotic vertebral fractures continues to be controversial. The most cited early randomized placebo-controlled trials from 2009 showed no clinically significant benefits of treatment with respect to pain, disability, or overall quality of life; however, those trials have some noted weaknesses and were lacking stringent patient selection criteria. When rigorous selection criteria are utilized, vertebral augmentation remains a treatment option that may improve pain, result in earlier hospital discharge, and reduce morbidity in selected patients.







