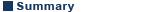The Chronicle of a Stainless Steel Orthopaedic Implant
Kempland C. Walley BcS, Mergim Bajraliu BcS, Tyler Gonzalez MD, MBA, Ara Nazarian PhD
The authors report no conflict of interest related to this work.
©2016 by The Orthopaedic Journal at Harvard Medical School
For years, orthopaedic surgeons have relied extensively upon stainless steel surgical fixation implants to treat patients with various levels of acute orthopaedic injuries. However, surgical implementation is merely one step in an otherwise intricate and complex process involving elemental stainless steel. This review, through illustration of the life-cycle of a stainless steel lower extremity fixation implant, aims to provide an understanding of the systems that govern the medical device industry. Prior to the arrival of a stainless steel implant to the operating room, there is a comprehensive product development process that closely assesses the various physical and chemical properties of the to-be implant. Beyond ensuring the inherent stability of the implant, developers must also establish compatibility of the implant within its destined anatomical environment. Furthermore, there are closely-regulated sterilization, packaging, and distribution protocols created to ensure the eventual safety and sustain the structural integrity of the implanted fixation device. In the case of hardware removal, these stainless steel surgical explants follow a poorly documented, uncertain, post-vivo fate, including - but not limited to - incineration and disposal, patient ownership, and passage to developing nations. Though seemingly simple, the life of stainless steel surgical fixation implants is surprisingly vast.
For orthopedic applications, surgical fixation implants commonly utilize stainless steel as a worthy candidate. This is largely due to its desirable structural properties, biocompatibility, and proven success in load bearing and fixation. Orthopedic surgeons use these devices routinely without giving thought to how these devices are designed, developed and engineered. The early life (i.e. development, manufacturing and implantation) of orthopedic-destined stainless steel is well understood, catalogued, and regulated until implantation. Once these implanted devices no longer become useful to the patient, following hardware removal for example, the fate of the implant is not as well catalogued. Because these implants are such an integral part of an orthopedic surgeon’s practice, it is not only interesting to consider their lifecycle, but also important for orthopedic surgeons to understand if questioned by a patient. This review aims to discuss the life span of orthopedic-destined stainless steel. Structurally, this review will segment this lifespan into three categories which span creation and development, implantation, and subsequest uses and fates of surgical fixation implants:
- Pre-vivo
- In-vivo
- Post-vivo
A shortened discussion on the in-vivo duration of an implant’s lifespan will be found in this review to confer more discussion on the other segments that are less commonly understood.
Material Selection & Product Development
In the development of any metal surgical device, material selection is a major criterion. Physical properties such as density, specific heat, melting point, thermal conductivity, thermal expansion, radiolucency, malleability and corrosion are thoroughly considered. The density of a metal is of particular importance for orthopedic applications. Increased density confers increased strength and stiffness – the ratio of strength-to-weight and stiffness-to weight, respectively. These are two fundamentally important concepts when considering the functionality of an implant. For instance, in the case of a hip replacement femoral stem, the biomaterial’s physical compliance should match to the adjacent bone to inhibit stress shielding and/or bone loss resulting from a lack of loading.
While the structural characteristics of metals and alloys make them suitable candidates for many load-bearing orthopedic applications, most metallic materials are intrinsically susceptible to chemical attack or corrosion from reacting with aqueous physiologic environments surrounding an injury.22 Implant degradation is not only concerning for the structural integrity of a device, but may also result in a systemic response in-vivo.4,16,22 As a result, materials chosen for implantation should be both resistant to oxidative or chemical stress and eligible for protective mechanisms such as passivation (i.e. the application of a thin coat of protective material or substance to create a shell against corrosion) to confer appropriate biocompatibility.11,22 For medical device product development engineers, the material selection process is a multifaceted responsibility factoring in geometry, structural and physiologic stresses and biological environment. Of the metal alloys that are used in fracture fixation plates, surgical tools, bone screws, dental implants, total joint replacements, and stents, candidate biocompatible metals include stainless steel, cobalt-chromium alloys, titanium and titanium alloys, and zirconium alloys.7,18,22
It is critically important for surgical device manufacturers to select a metal alloy that fares well in its destined anatomic location. Corrosion is heavily influenced by minute changes in pH; therefore local chemistry or coupling with another material can quickly turn a stable construct into a catastrophic one.4,17 For example, orthopedic hip stems are often made of titanium because of its excellent fatigue strength, light-weight, and its proven ability to reduce stress shielding, which can lead to localized bone loss surrounding the implant.17 Independent of adjacent metals, titanium forms an outer oxide layer that protects it from corrosion.11,17,22 However, in the presence of mechanical contact with other metals, this titanium oxide layer breaks down. This friction results in third-body particulate and wear of underlying titanium causing titanium alloys not to fare well under sliding contact applications that require load bearing, such as articulating joints.17,18,22 Other disadvantages of pure titanium include its low elastic modules when compared to stainless steel or cobalt-chromium (approximately one half that of stainless steel and cobalt-chromium), and low shear strength.4,11,22 Furthermore, titanium is markedly more difficult to manufacture and more expensive than stainless steel.
The primary stainless steel alloy recommended for surgical device manufacture is the American Iron and Steel Institute (AISI) type 316L, whereby the “L” designates low carbon content and 316 defines the metals grade as appropriate to be implanted, often called surgical stainless steel.11,22 While the composition of the alloy may vary slightly, type 316L stainless steel is derived from the commonly used 18-8 stainless steel alloy found in commercial products and tableware.14,17 This alloy is characterized by an 18-8% chromium and nickel content, respectively. Due to the need for heightened corrosion resistance, mainly to inter-granular attack and pitting, surgical implants require that molybdenum (3%) be introduced to the 18-8 alloy,24 and that carbon content is reduced (0.03% max). These compositional alterations motivate the addition of nickel (12%) to achieve the austenite microstructure (whereby austenite is defined as a solid solution of carbon in a nonmagnetic form of iron that stable at high temperatures). This structure is characterized by its face-centered crystal structure conferring a relative ease in shaping and bending, and superior ductility relative to the basic, body centered cubic crystal structure of Ferrite used in iron or low-alloy steel. The exact composition of this alloy is specified by ASTM International (formerly the American Society for Testing and Materials).4,17,22
While the mechanical properties of type 316L stainless steel make it a satisfactory biomaterial used in fixation implants, other biometals are also commonly used in orthopedic implants (i.e. Cobalt Chromium, Titanium).17,23 Nevertheless, stainless steel implants are numerous. Retrieval studies estimate that stainless steel alloys establish nearly 60% of the surgical implants used in the United States.2 Though stainless steel is commonly known for its corrosion resistance, it remains highly susceptible to stress corrosion, cracking and crevice corrosion.4,17 Consequently, its use is often limited to situations in which strength is not required for an extended duration.17
As the synthetic process of a metal can change a product’s inherent mechanical properties, it is important to assess whether an alloy is wrought or cast. A wrought alloy is worked by being forged or hammered, while a cast alloy describes a molten alloy that is poured into a mold to give it its desired shape. Wrought alloys are characterized by an annealed condition which retains better ductility than cast metal implants, which typically possess lower strength than those made by forging.17 Often, the wrought alloy undergoes thermal cycles in addition to manual forging to anneal the alloy, thereby enhancing its ductility. Since cast alloys are poured into molds, the resulting metal products are exceedingly more detailed and complex than possible through wroughting.8,20 Thus, casting alloys are usually the preferred selection in the manufacture of medical devices. This especially holds true with orthopedic fixation plates that require detailed, multifaceted contours to ensure functionality. Manufacture through casting bears significant economical savings.8
Given the composition and phase specifications of type 316 steel, cold-working (which is plastic deformation of metals below the recrystallization temperature, normally at room temperature) can also be employed.17,22 Cold-working not only hardens the steel to a two- to three-fold increase in yield strength, but it also generates a 40% increase in ultimate tensile strength.22 It also yields an 80% reduction in ductility. This net effect generates a far stronger material, but also makes it more brittle. Consequently, this hardening technique bears consideration when fabricating a fixation device, because as strength increases, the device becomes more susceptible to catastrophic failure: the point at which an implant irreversibly breaks.22
Forged stainless steel alloy is also commonly employed for fracture-fixation devices,25 since the forging process allows for the device to take on a stable crystalline structure. The most important reason for using forged stainless steel alloys is the selective directional deformation in forging a plate or fixation device from bar stock or in making wire.22 This results in an optimum “fiber texture” in which fibers are elongated into fibrous or spindle shapes. These fibrous spindles are parallel to the long axis of the device and the expected deforming forces, as indicated by the anatomical placement of the implant. Mechanically, this microstructure more effectively reduces the risk and severity of crack propagation and failure than do fully-annealed or as-cast devices having an equiaxed grain structure.22
Product development also incorporates an intricate computer modeling and proto-typing procedure. This process is termed finite element analysis. This practice offers a predictive, quantitative simulation of the complex biomechanics of the device. Furthermore, it offers valuable information on potential failure methods. Thus, computer modeling is imperative in the development of the implant.
Manufacture
Of the various biocompatible alloys currently used for implants, stainless steel is the most easily fabricated and the least expensive.25 Despite these advantages, yielding high quality stainless steel implants requires acute attention to the melting process, carbon content, and impurity content. Thorough assessment of the numerous thermal treatments is required to ensure detailed shaping and appropriate mechanical properties for the device’s clinical intention.8 Draw marks or scars, pits, burrs, and surface contamination must be eliminated by further surface preparation, such as mechanical or electro-polishing. Surface passivation in nitric acid is employed to remove unwanted surface iron particles. Passivation also aids in artificially thickening the surface oxide layer of the metal.8,22
Development
Contracting and Manufacture
Once a stainless steel implant has been designed to meet its functional requirements and has been vetted to manufacture, a medical device company typically enters an agreement with a contract manufacturer to carry out its production. Close attention must be paid to the manufacturing process to ensure that the intended elastic modulus is integrated within each implant. This ensures that proper load is transferred to adjacent tissues. Because bone healing is highly dependent on load control, it is imperative that the modulus and implant are properly assembled.17 After an order has been requested, the contract manufacturer requests the appropriate quantity of type 316L stainless steel metal in one of two forms:
- Flat stock
- Round stock
Flat stock stainless steel is typically used when machining fixation plates and is delivered to the manufacturer in rectangular form. Conversely, round stock metals are commonly utilized when machining surgical screws and are originally cylindrical. Specifications for both flat stock and round stock forms of stainless steel are highly regulated and standardized to ensure consistent quality in performance. Each manufacturer typically selects a particular alloy from an approved supplier list. Once the manufacturer has procured the alloy, there is a rigorous inspection of the mechanical properties, geometry, surface finish, overall straightness of the stock, and absence of physical deformities. Any imperfection in the alloy can potentially be attributed to a critically weak point in its microstructure.17 This can ultimately lead to a faulty final product.
Machining
After procurement and inspection, the alloy is machined into its penultimate form. There are three common machining techniques employed for most surgical implants: lathe machining, computer numerical control (CNC) Milling, and wire electro-discharge machining (EDM).9,17 Each technique corresponds to a specific implant type. Screw lathe machining, as the name implies, is commonly used for bone screws and components involving precise threading. Fixation plates and implants requiring complex geometry needed to match anatomical arches and grooves employ CNC High-Speed, Multi-axis Milling. This is due to the automated ability to simultaneously machine 5 faces of a part, generating compound angles and contours of any specification.9,10 CNC control for both of these modalities enables excellent precision, velocity and most importantly, reproducibility. Wire electrical discharge machining (EDM) is another technique used for applications requiring through-hole machining and fine work of metals that lack burrs.9,10 Wire EDM provides an efficient, non-contact and reproducible means of generating small, fragile, and intricate pieces that lack burrs. Despite the lengthy lead-time that is required to yield specific, geometries, batch processing - the execution of a series of commands, operations, or steps on a computer without manual intervention – can be invoked to reduce production times and reduce variability between outputs.9,10
Refinement and Passivation
Once machined to specification, a stainless steel implant undergoes a series of cleaning steps to remove residual pieces of adhering metal and unwanted material that might cause dislodging.17 The methods employed to clean recently machined stainless steel implants include fan blasting, rotary tumbling, hand polishing, centrifugation or a combination of the aforementioned techniques.
Following careful cleaning, surgical grade stainless steel implants undergo a passivation process that effectively removes free irons, thus improving corrosion resistance beyond the native properties of the metal.4,11,17,22 Passivation often utilizes nitric acid to not only remove surface iron particles, but also to thicken the surface oxide layer. As stainless steel has the lowest corrosion resistance among the most common metallic biomaterials, surface passivation is a vital process in manufacturing a stainless steel implant.17,22 This process ensures its biocompatibility. Implants with fine markings and details in particular necessitate sufficient passivation because of the increased surface area exposed to the bodily environment. Any method of passivation must conform to the procedures designated in the US standard ASTM 967A. Abiding by the designated procedure is generally required for achieving appropriate biocompatibility for a surgical implant. As metals are not inert and can spontaneously undergo galvanic corrosion, metal oxide layers can protect implants from corrosion potentiated from direct exposure with the electrochemical environment native to the human body.4,16,22 Following passivation, the implant should no longer be subjected to any action or process that might jeopardize the integrity of the passive layer.
Further refinement focuses on overall cleanliness or purity of the refined stainless steel alloy, which also influence the implant’s mechanical properties and vulnerability to corrosion. For implantation purposes, all steel alloys include impurities or inclusion bodies designed strategically to achieve a desired combination of functional and mechanical requirements and corrosion resistance.22 Experienced manufacturers may also recommend additional refinement to further improve implant performance. Generally, laser marking, fine engravings and metal finishing steps are carried out after passivation on stainless steel implants.17 For example, anodizing, an electrolytic passivation targeted to thicken the oxide layer produced, effectively enables the unique, dimensional color-coding of an orthopedic surgical kit.3,6,12 Electro-polishing, another metal finishing step, removes iron from the implant surface and increases the chromium/nickel content, which provides the most optimal form of passivation for stainless steel. Moreover, electro-polishing affords a clean, smooth, and bright surface on surgical implants.13,17 This not only makes them easier to sterilize, but also reduces any remaining micropeaks and valleys in the metal that could potentiate corrosion.17 A secondary washing step follows.
The approved use of an implant as a medical device requires a series of stringent validation and verification regulatory steps enforced by the Food and Drug Administration (FDA). These regulations ensure that the device confers desired performance specifications and is safe for medical use. Beyond those regulatory quality measures, most manufacturing shops adopt individualized labeling practices and itemized steps to assure that metal implants meet the geometric specifications outlined by the item’s blueprint.
Packaging & Distribution
Following careful inspection, the stainless steel surgical implant and its accompanying components are then packaged and prepared for distribution. It is common for future implants to have secondary and even tertiary packaging. In the case of a fracture fixation system kit, the series of fracture fixation plates and their corresponding screws are carefully organized into a metal case engineered for easy use in the operating room (Figure 1). This kit is then secured into another package with all necessary documentation (i.e. inspection reports, compliance report, recommended sterilization protocols, certifications of good manufacturing). The manufacturer will then aggregate all machined, cleaned, passivized, refined, cased and packaged surgical kits and ship them to the respective distribution channels outlined by each device company.
Once in the hospital
All medical devices and implants destined for human contact must be thoroughly clean and free of bacterial contamination. The definition of sterility is described as when less than one in one million surviving bacterial spores is on the device. This can be accomplished via autoclaving, irradiation, ethylene oxide gas (EtO), and gas plasma (Table 1). Generally, bio-metals can undergo any of these methods of sterilization because of their ability to withstand high temperatures. For most surgical kits, sterilization instructions are included in the packaging from the manufacturer. Thus, prior to implantation, the contents of the kits are sterilized in a health center’s sterilization facility according to the device’s appropriate sterilization method; most commonly via steam sterilization or autoclaving.26
Implantation
It is in the operating room where the first major step in the fate of a stainless steel implant takes place – by the discretion of the attending surgeon. The indication for this division depends on whether the implants destined for the patient fit the anatomical or pathological requirements of the injury and are fixated, or if the geometry of the part excludes it from implantation. If the device is implanted, the journey of the stainless steel implant continues parallel to its host throughout their lifetime, or until it is removed surgically. In the latter case, multiple avenues exist for the fate of stainless steel implants that are not immediately implanted. These include disposal, re-sterilization and subsequent storage, return to manufacturer, or collection and donation to developing nations.19,27
It is important to consider the interaction between the adjacent physiological environment and the orthopedic implant itself. More specifically, the exposed metal undergoes an electrochemical dissolution from exposure to extracellular tissue fluid at a finite rate.28 When a fixation plate is implanted the local tissue environments are disturbed due to mechanical compression of the bone and resultant compression of the periosteal vasculature, causing a disruption in blood supply to the surrounding tissue and also altering existent homeostatic ionic equilibrium. With 316L stainless steel, a naturally corrosive biomaterial, iron, chromium and nickel ions are passively released over time –which are believed to have allergic and carcinogenic properties in-vivo.23 Corrosion may also lead to physical changes in an implant heightening the likelihood of mechanical failure. It is theorized that 90% of 316L stainless steel surgical implant failures are the result of pitting (cavities or “holes” produced resulting from the aggregation of localized electrochemical cells) and crevice corrosion (the loss of material at the coupling interface between a plate and a locking screw).29 Other methods of corrosion include inter-granular corrosion, fretting, galvanic corrosion and stress corrosion cracking. These unstable pathways, combined with the natural corrosive properties of metallic implants, potentiate the risk of implant failure or may trigger immune-mediated rejection of the implant.30 In these cases, the implant will be promptly removed.
Implant removal is characterized as one of the most common elective orthopedic procedures, contributing to an estimated 30% of all planned orthopedic cases.14 In adults, indications for implant removal include local pain, soft tissue irritation, infection, ligamentous and tendon obstruction and (while not usually the case for fixation plates) the return to rigorous activity or sports following healing with syndesmotic screws. In children, it is considered reasonable to remove implants immediately after healing to sidestep possible conflicts to the growing skeleton. This is done to prevent immuring, which would make later removal extremely difficult. Furthermore, this permits future reconstructive procedures following bony maturation (e.g., Proximal femur locking plates for proximal femur rotational osteotomies).31 In the case of a stainless steel implant, avoiding corrosion and its presumed toxic, allergic or potential carcinogenic potential motivate implant removal. Implant removal, however, necessitates a second surgical exposure in scarred tissue, and poses a risk for nerve damage, re-fractures and infection.15,21,32,33 Despite the risks and benefits of hardware removal, there is no consensus on routine removal of hardware from well-healed fractures. The current orthopedic literature concerning re-fracture risk from retained implants neither supports nor refutes universal retention or removal of hardware, calling for additional factors to be considered when hardware removal may be indicated. These considerations include, but are not limited to, local pain, fixation across joints, metal allergy, carcinogenicity, pediatric patients, and surgical complications.5 Moreover, no clear guidelines have been established defining how long patients should refrain from load bearing after hardware removal.5 Nonetheless, hardware removal embodies a common procedure in orthopedic surgery and consideration of the final destiny of these implants is warranted in this chronicle as the life of a stainless steel implant does not simply end once removed from a patient.
There is no consensus on the eventual fate of the implant following removal. Some orthopaedic surgeons speculate that once a stainless steel implant is removed from a patient, it is shipped to pathology and subsequently recycled. Others believe that systems are in place within medical centers to monetize the harvested material. There is also speculation that implants are donated to developing countries for secondary use. In fact, harvested implants frequently become collector items by attending physicians, residents and fellows. In some instances, removed implants are given back to the patient as a gift.
Why is there a lack of understanding of what happens?
There are a few potentially logical fates for a fixation implant following removal, regardless of functional intactness. From a quality perspective, it may make sense if all explants were biomechanically evaluated by the source company to determine how its material properties relate to its performance prior to implantation. Similarly, the source company could assess how the implant’s passivation process actually holds up in-vivo. Though logical, this practice is seldom undertaken from anecdotal commentary from individuals the authors have contacted who could adequately speak to this practice. Presumably, numerous companies have investigated this; however, there is no indication that this practice will become an industry standard. There is only one circumstance that necessitates the return of the explant to the manufacturer. Otherwise, an explant’s fate is left to the discretion of the institution conducting the hardware removal. For instance, the Association of Perioperative Registered Nurses (AORN) recommends that policies regarding the disposition of surgical specimens and explants are established by a multidisciplinary organization. They advise that representatives from various departments, including the Pathology Department, facility or health care organization physicians, and perioperative registered nurses should come to a consensus in accordance with local, state, and federal regulations.19 Moreover, the College of American Pathologists (CAP) encourages each health care organization to establish a written policy that documents, which explanted specimens, are exempt from pathology examination, and which specimens should undergo gross examination but are exempt from microscopic examination. This association also suggests that each health care organization delineate when certain explanted specimens be exempt from pathological examination provided there is an established documentation and disposition procedure in place, and that certain specimens undergo gross examination under the discretion of the corresponding pathologist.27
While there is no universal standard or regulation on how explanted orthopedic devices should be collected and handled, there are elementary guidelines in place should the implant be returned to the patient, provided that it is an acceptable practice at that health care facility. These guidelines include decontamination, labeling, packaging and documentation established by the health care organization policies and procedures. This, however, is completely discretionary.
In our level one, urban academic trauma institution, however, the majority of explanted orthopedic devices are not submitted to pathology, returned to the manufacturer, or returned to the patient. Rather, they are disposed of in the same fashion as the disposal of sharps – local aggregation and eventual incineration. Metal implants sent to pathology for examination have the same destiny. While this seems to be the end of the road for stainless steel orthopedic implants in the United States, and presumably in other leading medical countries in the world, the chronicle, in fact, may continue in developing countries.
There is a lack of literature describing the practice of orthopedic implant transport and integration into healthcare structures in developing countries. Given the lack of regulation in this area, this comes as no surprise.
To close this gap in knowledge, it is possible to query various orthopedic chairs and chiefs of orthopedic specialty services in academic medical centers that engage in this practice (directly or peripherally) and abroad who engage in volunteer work in developing countries from multiple specialties (i.e. Trauma, Hand and Upper Extremity, Foot and Lower Extremity, Spine) to provide anecdotal accounts surrounding fixation implants.
One might expect that implanted materials are never replanted (secondary use) in patients, regardless of their perceived acceptable condition. While unlikely, it is plausible that in dire, life-threatening circumstances, a clean and readily available secondary implant might be used to promptly reduce a fracture to avoid fatal internal bleeding. As part of a thorough investigation, it is possible to discover that inventories of explanted hardware, including plates, screws, and nails are kept near operating rooms in low-resource, medical centers abroad. The contents of these inventories, theoretically, can be sterilized and ready for implantation, should a situation demand their use.
It is interesting to consider sterilization for implants that reach developing countries, regardless of how they arrived, In areas without access to proper sterilization equipment, surgical instruments are, at best, disinfected with chemicals or boiling water prior to implantation.34 Failure to properly sterilize devices destined for contact with sterile tissue may result in infection or mortality.1 Simply using disinfectants does not eradicate all bacterial spores.35 It has, however, been suggested that the use of liquid chemical sterilants can be a reliable method of sterilization for surgical implants provided all organic and inorganic surface material is eliminated, and if the proper specifications for concentration, contact duration, temperature and pH are followed precisely.19,36 Despite these criteria, since devices cannot be properly wrapped during processing in the chemical sterilant, maintaining sterility immediately after processing or storage is near impossible.
While perhaps unsatisfying when compared to the plentitude of information available describing the pre-vivo and in-vivo segments of a surgical implants lifespan, the final chapter of this review should highlight the need for further investigation and surveillance of what truly happens following explanation and prompt a discussion amongst those who have engaged in the delivery or practice of explanted orthopedic devices.